Immune biomarkers for diagnosis of Axial Spondyloarthritis
Spondyloarthritis (SpA) is a group of rheumatic diseases with a heterogeneous clinical presentation, which can be classified as axial (ax) and peripheral SpA.
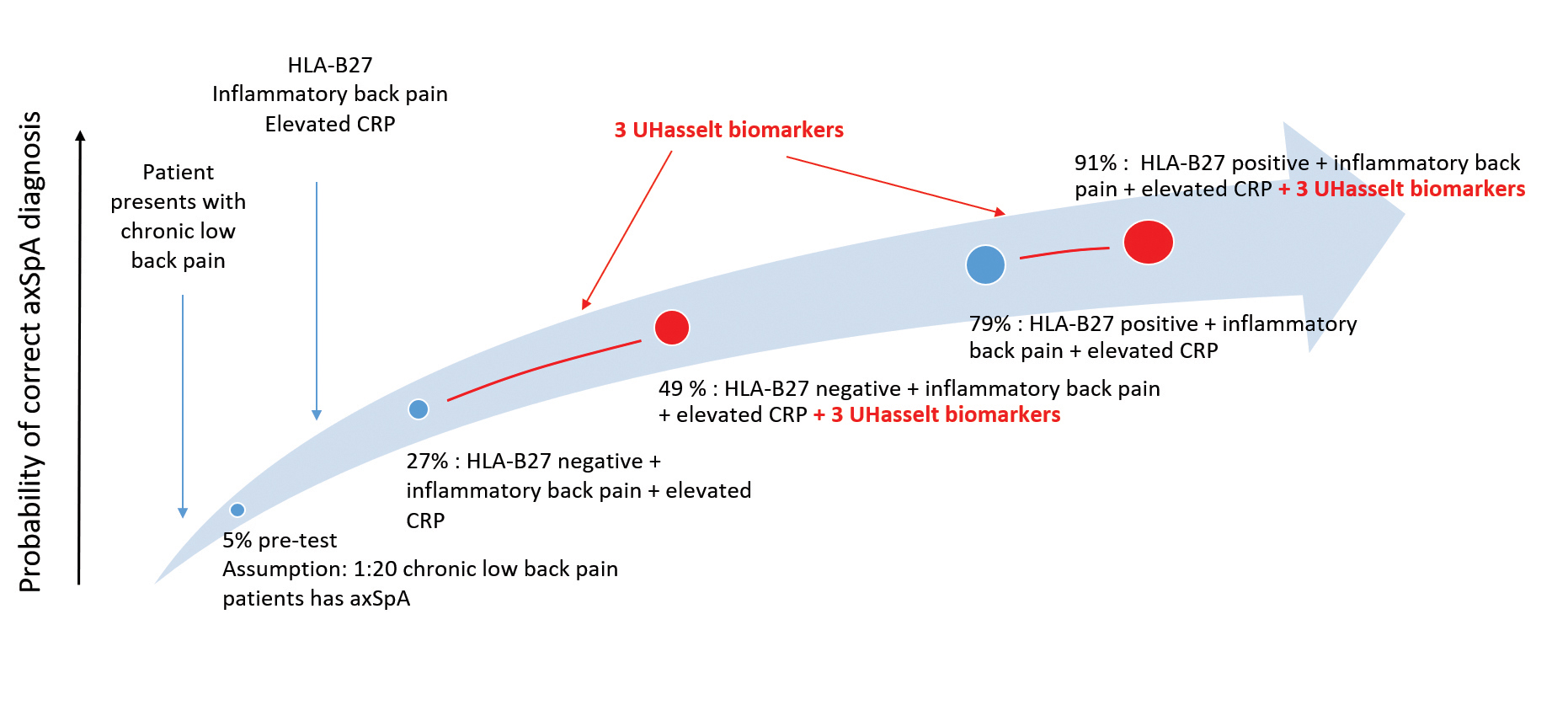
AxSpa, predominantly characterized by inflammatory back pain, has an estimated prevalence of 0.7%. However, due the lack of an appropriate serological test, there is a diagnostic delay of 5-10 years. Moreover, at present it is challenging to distinguish between axSpA patients and persons with chronic low back pain at an early stage, as diagnostic criteria are lacking.
Hasselt University and KU Leuven have discovered and validated 3 new biomarkers in plasma/serum for diagnosis of axSpA.


COMPELLING RESULTS
The 3 UHasselt biomarkers have a sensitivity of 14% and a corresponding specificity of 95% in chronic low back pain patients.
Combined with the presence of inflammatory back pain, and the currently used laboratory markers HLA-B27 and CRP, the 3 UHasselt biomarkers increase the probability of correct axSpA diagnosis
° from 79% to 91% for HLA-B27 positive patients with inflammatory back pain and elevated CRP;
° from 27% to 49% for HLA-B27 negative patients with inflammatory back pain and elevated CRP;
KEY FEATURES AND ADVANTAGES
Diagnostic value of 3 biomarkers, validated in 2 independent cohorts (n=260).
Research ELISA assays translated to high throughput peptide ELISA assays.
Possibility to better diagnose axSpA patients and start correct treatment earlier.
Hasselt University and KU Leuven have a strong IP protection for the biomarkers.
MARKET POTENTIAL
With a population prevalence between 0.3 - 1.4% and a diagnostic prevalence of 0.2 - 0.7%, better axSpA diagnosis will have a large potential.
While treatment of axSpA has been largely improved in the last decennia, diagnostics is still very limited. With a compound annual growth rate (CAGR) of 7% for the therapeutic market, better and earlier diagnosis is key.
Using our biomarkers, axSpA patients can be diagnosed better and earlier, resulting in earlier and correct treatment.
OUTSTANDING OPPORTUNITY
Patent application is available for licensing.
Both universities are searching for interested parties to complete development and commercialization.
RELEVANT PUBLICATION
Quaden et al., Arthritis Rheumatol. 2020 Jul 8. doi: 10.1002/art.41427
BUSINESS DEVELOPER
dr. Leen Willems

Do you want this information in a pdf? Click here.
